45 drug warning labels examples
› drugs › drug-safety-and-availabilityFDA identifies sudden discontinuation of opioid pain medicines Examples of common opioids include codeine, fentanyl, hydrocodone, hydromorphone, morphine, oxycodone, and oxymorphone. Health care professionals should not abruptly discontinue opioids in a ... FDA Drug Safety Communication [ 12-14-2016 ] The U.S. Food and Drug Administration (FDA) is warning that repeated or lengthy use of general anesthetic and sedation drugs during surgeries or procedures in children younger than ...
› tobacco-products › productsE-Cigarettes, Vapes, and other Electronic Nicotine Delivery ... Get an overview of FDA regulation of vapes, e-cigarettes, and other electronic nicotine delivery systems. You can also find statistics about current use.

Drug warning labels examples
› drugs › drug-safety-and-availabilityFDA Drug Safety Communication: Prescription Acetaminophen ... In addition, a Boxed Warning highlighting the potential for severe liver injury and a Warning highlighting the potential for allergic reactions (e.g., swelling of the face, mouth, and throat ... FDA Drug Safety Communication: FDA warns about serious heart … [06-07-2016] The U.S. Food and Drug Administration (FDA) is warning that taking higher than recommended doses of the common over-the-counter (OTC) and prescription diarrhea medicine loperamide ... Radio-frequency identification - Wikipedia Radio-frequency identification (RFID) uses electromagnetic fields to automatically identify and track tags attached to objects. An RFID system consists of a tiny radio transponder, a radio receiver and transmitter.When triggered by an electromagnetic interrogation pulse from a nearby RFID reader device, the tag transmits digital data, usually an identifying inventory number, …
Drug warning labels examples. › food › food-labeling-nutritionChanges to the Nutrition Facts Label | FDA - U.S. Food and ... Mar 07, 2022 · Manufacturers with $10 million or more in annual sales were required to update their labels by January 1, 2020; manufacturers with less than $10 million in annual food sales were required to ... Pet Food Labels - General | FDA - U.S. Food and Drug … In these examples, at least 95% of the product must be the named ingredient (beef or tuna, respectively), not counting the water added for processing and "condiments." Counting the added water ... › drugs › drug-safety-and-availabilityFDA Drug Safety Communication: FDA warns about serious heart ... [06-07-2016] The U.S. Food and Drug Administration (FDA) is warning that taking higher than recommended doses of the common over-the-counter (OTC) and prescription diarrhea medicine loperamide ... FDA Drug Safety Communication: FDA strengthens warning … [FDA strengthens warning that non-aspirin nonsteroidal anti-inflammatory drugs (NSAIDs) can cause heart attacks or strokes (FDA Drug Safety Communication). Based on FDA’s comprehensive review of ...
E-Cigarettes, Vapes, and other Electronic Nicotine Delivery … Get an overview of FDA regulation of e-cigarettes, vapes, and other electronic nicotine delivery systems. You can also find statistics about current use. FDA identifies sudden discontinuation of opioid pain medicines In turn, these symptoms can lead patients to seek other sources of opioid pain medicines, which may be confused with drug-seeking for abuse. Patients may attempt to treat their pain or withdrawal ... Changes to the Nutrition Facts Label | FDA - U.S. Food and Drug ... 07.03.2022 · Examples would be a 24-ounce bottle of soda or a pint of ice cream. With dual-column labels available, people can more easily understand how many calories and nutrients they are getting if they ... FDA Drug Safety Communication: FDA cautions about using … [03-03-2015] The U.S. Food and Drug Administration (FDA) cautions that prescription testosterone products are approved only for men who have low testosterone levels caused by certain medical ...
› drugs › drug-safety-and-availabilityFDA Drug Safety Communication: FDA cautions about using ... Testosterone replacement therapy is only approved for men who have low levels of testosterone related to certain medical conditions. Examples of these conditions include genetic problems, and ... Common Grapefruit Juice Drug Interactions - Drugs.com 18.05.2022 · While grapefruit is a nutritious fruit, many patients are concerned about the potential for drug interactions with grapefruit juice. Maybe you've receive a medication prescription container with an affixed warning label that recommends you avoid grapefruit or grapefruit juice while taking the medication. Grapefruit juice interactions can even ... › drugs › drug-safety-and-availabilityFDA Drug Safety Communication: FDA review results in new ... [ 12-14-2016 ] The U.S. Food and Drug Administration (FDA) is warning that repeated or lengthy use of general anesthetic and sedation drugs during surgeries or procedures in children younger than ... Radio-frequency identification - Wikipedia Radio-frequency identification (RFID) uses electromagnetic fields to automatically identify and track tags attached to objects. An RFID system consists of a tiny radio transponder, a radio receiver and transmitter.When triggered by an electromagnetic interrogation pulse from a nearby RFID reader device, the tag transmits digital data, usually an identifying inventory number, …
FDA Drug Safety Communication: FDA warns about serious heart … [06-07-2016] The U.S. Food and Drug Administration (FDA) is warning that taking higher than recommended doses of the common over-the-counter (OTC) and prescription diarrhea medicine loperamide ...
› drugs › drug-safety-and-availabilityFDA Drug Safety Communication: Prescription Acetaminophen ... In addition, a Boxed Warning highlighting the potential for severe liver injury and a Warning highlighting the potential for allergic reactions (e.g., swelling of the face, mouth, and throat ...








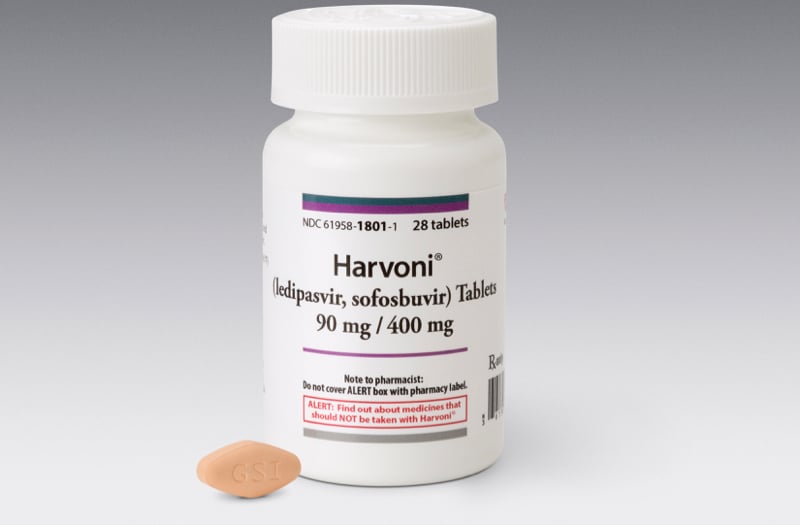









![PDF] Improving prescription drug warnings to promote patient ...](https://d3i71xaburhd42.cloudfront.net/e087ba4ebaa8ed043bf22bd2ab63292d6a05b9d4/2-Figure1-1.png)








![PDF] Improving prescription drug warnings to promote patient ...](https://d3i71xaburhd42.cloudfront.net/e087ba4ebaa8ed043bf22bd2ab63292d6a05b9d4/3-Figure2-1.png)


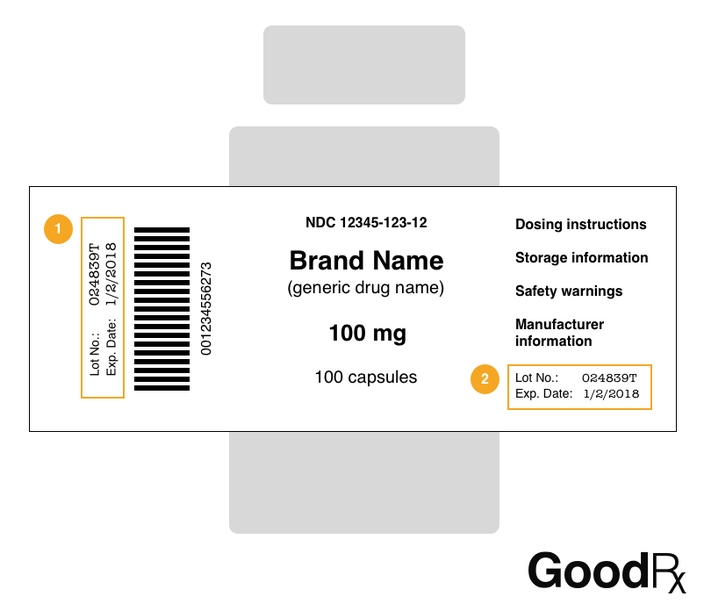






Post a Comment for "45 drug warning labels examples"